Overview
Our product safety is ensured by the most rigid safety measures among existing scientific standards.


Description
This product is a light yellow-brown or yellow-brown clear liquid with a distinctive odor. The pH level ranges from 5.5 to 6.5 and the osmotic pressure ratio (to physiological saline) is approximately one.
Indications
Improvement of hepatic function in chronic hepatic disease.
Dosage and administration
The normal adult dose is a 2 ml subcutaneous or intramuscular injection once daily. According to symptoms, the dose can be increased to 2 or 3 times daily.
Packaging
2 ml 50 ampoules
Cautions for use**
1. Careful Administration
LAENNEC should be administrated with care in patients predisposed to allergies.
2. Important Basic Cautions
This product is manufactured from the extract of human placenta delivered full-term in Japan. In order to screen each donor, a complete medical history, interviewing such as a history of travel and serologic testing for viruses, bacteria and infection are performed, after nucleic-acid testing (NAT) to meet with requirements for HBV-DNA, HCV-RNA and HIV-1-RNA is carried out. In addition, it has been confirmed that high-pressure steam sterilization for 20 minutes at 121 °C during the manufacturing process is effective in inactivating various viruses such as HIV etc. Furthermore, although in the product test, the nucleic-acid test meets with requirements for HBV-DNA, HCV-RNA, HIV-1-RNA, HTLV-DNA, and parvovirus B19-DNA, patients should be made aware of the following points during administration: To date, the transmission of infection, such as variant Creutzfeldt-Jakob disease (vCJD), by administration of this product in Japan or other countries has not been reported. However, although safety measures are taken to prevent infection during the manufacturing process, it is theoretically impossible to fully eliminate the risks of infection transmission originating in a human placenta used as raw material. While safety measures during the manufacturing process to prevent infection, as well as confirmation of the necessity of treatment for the disease before administration are carried out, doctors should explain to patients and try to have them understand that when human placenta is used as the raw material of a product, the risk of infection cannot be fully ruled out.
3. Drug Interactions
When this product is directly mixed with a strong base preparation of pH 8.5 or more, attenuation of pharmacological activity has been reported.
No coadministration with this product resulting in the enhancement or attenuation of the pharmacological effect of this product or concomitant drugs, appearance of adverse reactions, or aggravation of disease has been reported.
Adverse reactions or patients who were suspected to have suffered adverse reactions to this product were reported in a total of 10 (3.7 %) of the 273 patients selected for safety evaluation in the clinical study performed during implementation of reevaluation of the drug efficacy. The most frequently observed adverse reactions were injection site pain in 7 patients (2.6 %), hypersensitivity (such as rash, fever, and itching) in 1 patient (0.4 %), injection site indurations in 1 patient (0.4 %), and gynaecomastia in 1 patient (0.4 %). The causal relationship between gynaecomastia and this product is unknown.
No abnormal changes in laboratory values were observed.1)
• Clinically significant adverse reactions
**Shock (incidence unknown):
Since this product is a protein/amino acid preparation derived from human tissue, this product may cause a shock. If any signs of abnormality are observed, the drug should be discontinued immediately, appropriate measures should be taken and the condition should be monitored fully.
• Other adverse reactions (in descending order of occurrence)
| Injection site pain | 2.56% |
| Sensitivity (rash, fever, itching etc.) | 0.37% |
| Injection site indurations | 0.37% |
| Gynaecomastia | 0.37% |
4. Use in the Elderly
From clinical data and present use, no particular caution is needed in the administration of this product to the elderly. However, since elderly patients often have reduced physiological function, it should be administrated with care.
5. Use during Pregnancy. Delivery or Lactation
In reproductive development toxicity experiments on animals, including teratogenicity, this product can be considered not to have such toxicity.2)
6. Pediatric use
The safety of this product in premature infants, newborns, infants, toddlers or children has not been established due to insufficient clinical data.
7. Overdosage
Overdosage of this product and the resulting efficacy or safety have not been established (Due to insufficient clinical data).
8. Precautions for Use
Injection site:
In order to avoid any effect on tissue or nerves, the product should be injected subcutaneously or intramuscularly taking the following precautions:
a) For the injection site, to avoid nerve pathways, it should be administrated with care.
b) In the case of repeated injections, avoid injecting into the same site by alternating on the left and right side etc.
c) If intense pain or regurgitation of blood is observed, the needle should be removed immediately, and injected into a different site.
Opening the ampoule:
When opening the ampoule, it is preferable to wipe the part to be cut with an ethanol sponge before opening it.
Pharmacokinetics
The bioactive ingredients of LAENNEC are extracted from human placenta, and the primary pharmacological effects of this product can not necessarily be attributed to a single substance or ingredient. Therefore, evaluation of the pharmacokinetics (absorption, distribution, metabolism, and excretion) of this product has not been established.
Clinical studies
1. Double blind comparison to chronic hepatitis and hepatic cirrhosis1)
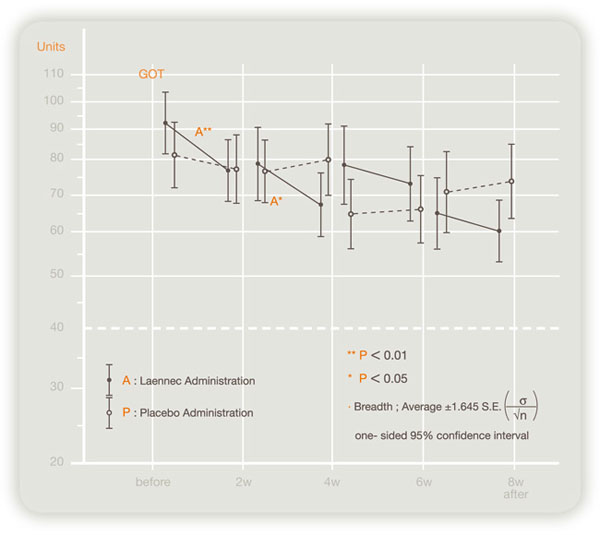
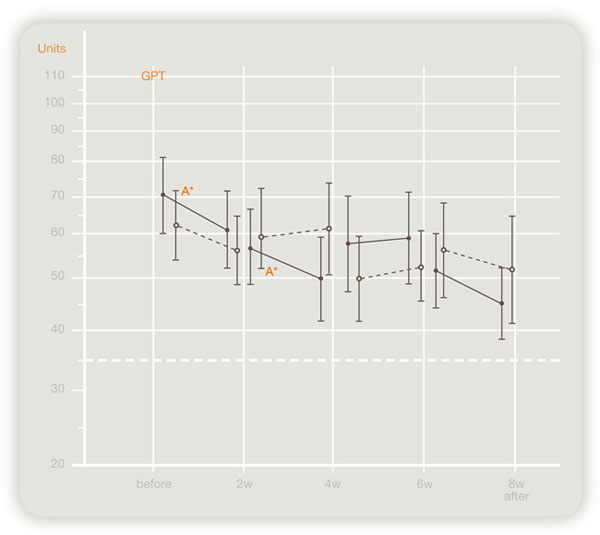
In a double-blind crossover study of 124 patients in Japan on the effects of this product on chronic hepatitis and hepatic cirrhosis, administration of this product improved serum transaminase (GOT, GPT) levels significantly (see figures below).
Efficacy to GOT
Efficacy to GPT
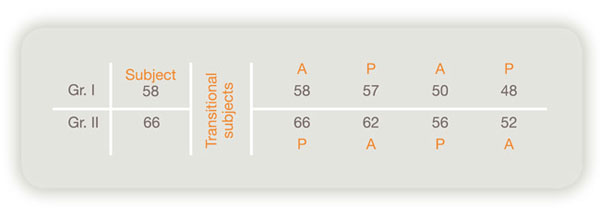
Summary of drug-efficacy [(A) Items having a tendence to decrease or increase significantly by Laennec]
More Features

Castillo's Global Partnerships and Diverse Operations
Be our partner in success
Contact Us
Looking For
A Special Offers?
Castillo International General Trading And Contracting Company WLL And Its Associate Companies Are A Kuwaiti Company Operating In Several Fields, And Have Activities In The Countries Of The Gulf Cooperation Council.
- General Trading And Contracting
- Iron Trade - Iron Contracting - Steel Pipes
- Selling Ready-Made Tubes And Accessories
Reach To Us
- Our Office : Kuwait City – Sharq- Block 5 – Ahmad Al-Jaber Street – Hamad Tower – Floor 12.
- Our Email : Info@Castillo.Com.Kw
- Call : +965 66866702 (Call For Enquiries)

